HOW ZYTIGA® (abiraterone acetate) WORKS
ZYTIGA® (abiraterone acetate) in combination with prednisone is indicated for the treatment of patients
- with metastatic castration-resistant prostate cancer (mCRPC)
- with metastatic high-risk castration-sensitive prostate cancer (CSPC)
MECHANISM OF ACTION
Abiraterone acetate (ZYTIGA®) is converted in vivo to abiraterone, an androgen biosynthesis inhibitor, that inhibits 17α-hydroxylase/C17,20-lyase (CYP17). This enzyme is expressed in testicular, adrenal, and prostatic tumor tissues and is required for androgen biosynthesis.
There are multiple sources of androgen, including the testes and adrenal glands. However, in metastatic prostate cancer, the tumor itself can be an additional source of androgen production.
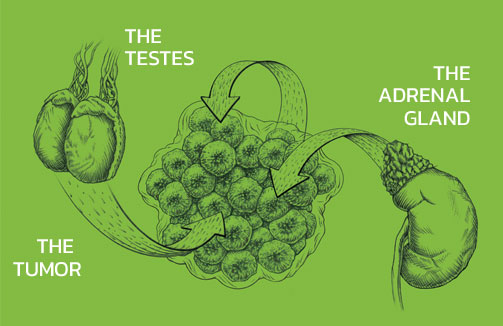
ZYTIGA® inhibits androgen production at 3 sources: the testes, the adrenal glands, and the prostate tumor itself.
Hypokalemia, Fluid Retention, and Cardiovascular Adverse Reactions Due to Mineralocorticoid Excess - ZYTIGA® may cause hypertension, hypokalemia, and fluid retention as a consequence of increased mineralocorticoid levels resulting from CYP17 inhibition [see Clinical Pharmacology (12.1)]. Monitor patients for hypertension, hypokalemia, and fluid retention at least once a month. Control hypertension and correct hypokalemia before and during treatment.
Closely monitor patients whose underlying medical conditions might be compromised by increases in blood pressure, hypokalemia, or fluid retention, such as those with heart failure, recent myocardial infarction, cardiovascular disease, or ventricular arrhythmia. In postmarketing experience, QT prolongation, and torsades de pointes have been observed in patients who develop hypokalemia while taking ZYTIGA®.
The safety of ZYTIGA® in patients with left ventricular ejection fraction <50% or New York Heart Association (NYHA) Class III or IV heart failure (in COU-AA-301) or NYHA Class II to IV heart failure (in COU-AA-302 and LATITUDE) has not been established because these patients were excluded from these randomized clinical trials [see Clinical Studies (14)].
Learn more about the efficacy and safety of ZYTIGA® (abiraterone acetate)


